Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (6): 912-918.doi: 10.3969/j.issn.2095-4344.2014.06.015
Previous Articles Next Articles
Mononuclear cells isolated from mobilized bone marrow differentiate into vascular endothelial cells and cardiomyocyte-like cells
Yao Wei1, Zhang Xiu-lan1, Yao Ying2, Wang Wei-shu1
- 1Second Affiliated Hospital of Shanxi Medical University, Taiyuan 030001, Shanxi Province, China; 2Department of Geriatrics, First Affiliated Hospital of Shanxi Medical University, Taiyuan 030001, Shanxi Province, China
-
Revised:2013-12-05Online:2014-02-05Published:2014-02-05 -
About author:Yao Wei, M.D., Associate professor, Second Affiliated Hospital of Shanxi Medical University, Taiyuan 030001, Shanxi Province, China -
Supported by:the Tackle Key Program of Shanxi Provincial Health Bureau, No. 200738
CLC Number:
Cite this article
Yao Wei, Zhang Xiu-lan, Yao Ying, Wang Wei-shu. Mononuclear cells isolated from mobilized bone marrow differentiate into vascular endothelial cells and cardiomyocyte-like cells[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(6): 912-918.
share this article
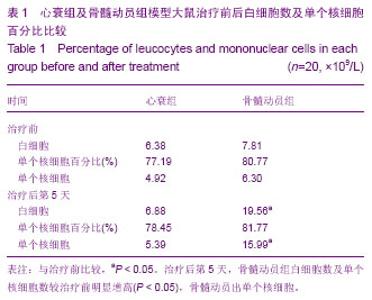
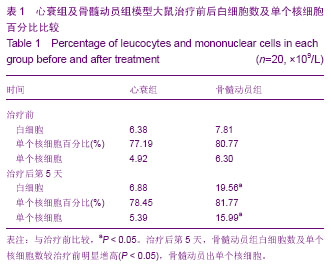
2.1 实验动物数量分析 纳入80只大鼠制备阿霉素诱导心肌病心衰模型,50只符合造模标准,分为2组,骨髓动员组30只,心衰组20只,全部进入结果分析,无脱失。 2.2 各组大鼠治疗前后外周血白细胞总数及单个核细胞百分比 见表1。 骨髓动员组治疗后第5天较治疗前白细胞数明显增高,差异有显著性意义(P < 0.05);单个核细胞百分比无明显增高,差异无显著性意义(P > 0.05)。因此骨髓动员组根据白细胞明显增高,计算出单个核细胞(单个核细胞=白细胞×单个核细胞百分比),在骨髓动员组治疗后第5天较治疗前明显增高,差异有显著性意义(P < 0.05)。心衰组治疗后第5天较治疗前白细胞无明显增高,差异无显著性意义(P > 0.05);单个核细胞百分比无明显增高,差异无显著性意义(P > 0.05)。因此心衰组根据白细胞无明显增高,计算出单个核细胞在心衰组治疗后第5天较治疗前无明显增高,差异无显著性意义(P > 0.05)。这充分说明骨髓动员出单个核细胞。"
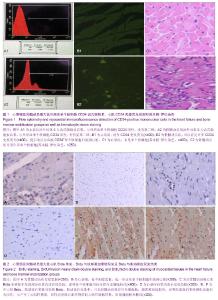
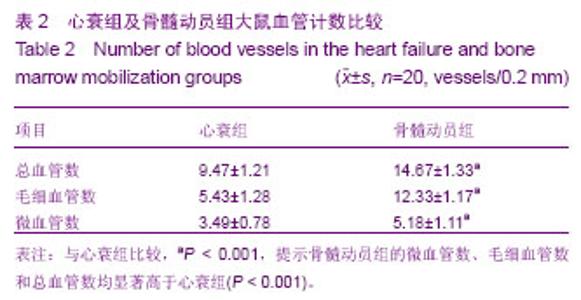
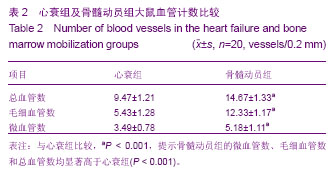
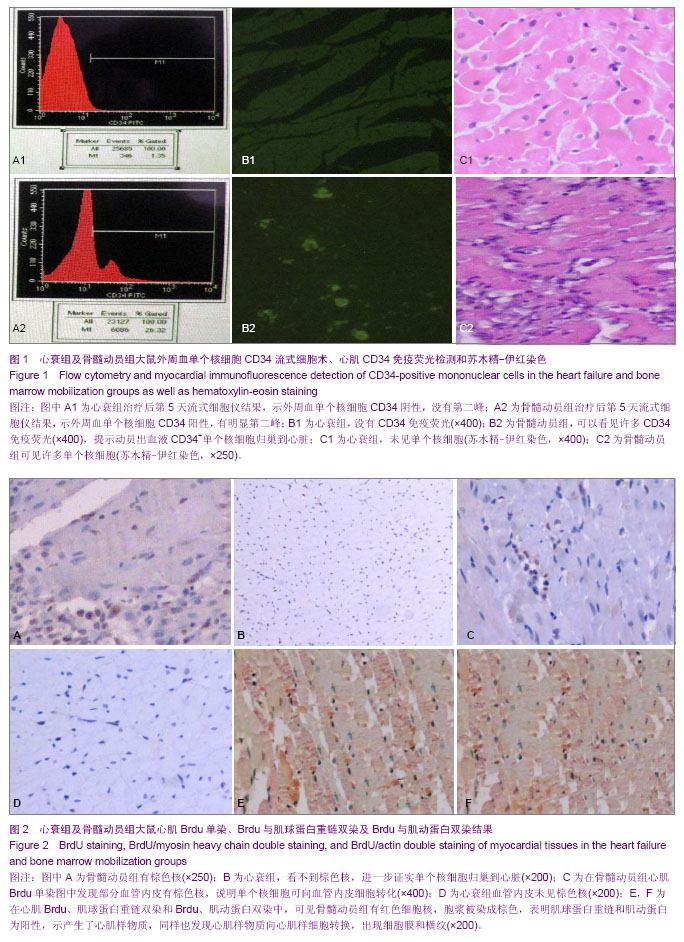
2.3 治疗后第5天模型大鼠外周血CD34水平 通过流式细胞仪检测,骨髓动员组的CD34为阳性,有明显第二峰;而心衰组为阴性,没有第二峰。骨髓动员组与心衰组大鼠外周血单个核细胞CD34阳性率分别为(26.35±0.91)%,(1.41± 0.77)%,骨髓动员组高于心衰组,差异有显著性意义(P > 0.05),见图1A。 2.4 模型大鼠心肌CD34免疫荧光检测结果 通过心肌CD34免疫荧光可以看出心衰组没有CD34免疫荧光,为阴性(图1B1);而骨髓动员组可以看见许多CD34免疫荧光,为阳性(图1B2),因此可认为动员出血液CD34+单个核细胞归巢到心脏。 2.5 模型大鼠心肌切片苏木精-伊红染色结果 见图1。 心衰组可见部分心肌细胞肥大,心肌细胞的胞浆内有空泡形成、间质水肿、心肌细胞间隙增宽、心肌纤维断裂和肌原纤维溶解,没有似炎性细胞,且又不是心肌细胞的单个核细胞(图1C1)。 骨髓动员组可见心肌细胞的修复,看到许多类似炎性细胞,且又不是心肌细胞的单个核细胞(图1C2),而心衰组切片苏木精-伊红缺如,推测就是骨髓动员出的CD34+单个核细胞。 2.6 模型大鼠心肌Brdu单染结果 通过心肌Brdu免疫组化染色,可见骨髓动员组有棕色核(图2A),而心衰组为阴性(图2B),看不到棕色核,进一步证实了单个核细胞归巢到心脏。 2.7 模型大鼠单个核细胞分化为血管内皮细胞及血管计数 在骨髓动员组心肌Brdu单染图中发现部分血管内皮有棕色核,说明单个核细胞可向血管内皮细胞转化。在苏木精-伊红染色下各组血管数经统计学结果显示,骨髓动员组的微血管数、毛细血管数和总血管数均显著高于心衰组 (P < 0.001),见图2C,D及表2。 2.8 心肌Brdu与肌球蛋白重链双染及Brdu与肌动蛋白双染 在心肌Brdu、肌球蛋白重链双染和Brdu、肌动蛋白双染中,可见骨髓动员组有红色细胞核,说明单个核细胞归巢到心脏,同时也看到胞浆被染成棕色,表明肌球蛋白重链和肌动蛋白为阳性,示产生了心肌样物质,同样也发现心肌样物质向心肌样细胞转换,肌样细胞分化,出现细胞膜和横纹。此外也可见一些红色细胞核但胞浆未被染成棕色,说明单个核细胞尚未产生了心肌样物质(图2E,F)。 "
| [1] 肖文涛,高丽君,高传玉,等.不同类型骨髓干细胞治疗扩张型心肌病的疗效对比[J].中华心血管病杂志,2012,40(7):575-577.[2] Arnous S, Mozid A, Mathur A. The Bone Marrow Derived Adult Stem Cells for Dilated Cardiomyopathy (REGENERATE-DCM) trial: study design. Regen Med. 2011;6(4):525-533.[3] Yeo C, Mathur A. Autologous bone marrow-derived stem cells for ischemic heart failure: REGENERATE-IHD trial. Regen Med. 2009;4(1):119-127.[4] Möbius-Winkler S, Höllriegel R, Schuler G, et al. Endothelial progenitor cells: implications for cardiovascular disease. Cytometry A.2009;75(1):25-37.[5] Brunner S, Engelmann MG, Franz WM. Stem cell mobilisation for myocardial repair. Expert Opin Biol Ther. 2008;8(11): 1675-1690.[6] Li TS, Takahashi M, Ohshima M, et al. Myocardial repair achieved by the intramyocardial implantation of adult cardiomyocytes in combination with bone marrow cells. Cell Transplant. 2008;7(6):695-703.[7] Cheng Z, Liu X, Ou L,et al. Mobilization of mesenchymal stem cells by granulocyte colony-stimulating factor in rats with acute myocardial infarction. Cardiovasc Drugs Ther. 2008; 22(5):363-371. [8] De Silva R, Raval AN, Hadi M, et al. Intracoronary infusion of autologous mononuclear cells from bone marrow or granulocyte colony-stimulating factor-mobilized apheresis product may not improve remodelling, contractile function, perfusion, or infarct size in a swine model of large myocardial infarction. Eur Heart J. 2008;29(14):1772-1782.[9] Ripa RS, Kastrup J. G-CSF therapy with mobilization of bone marrow stem cells for myocardial recovery after acute myocardial infarction --a relevant treatment? Exp Hematol. 2008;36(6):681-686.[10] Brunner S, Huber BC, Fischer R, et al.G-CSF treatment after myocardial infarction: impact on bone marrow-derived vs cardiac progenitor cells. Exp Hematol. 2008;36(6):695-702. [11] Inoue T, Sata M, Hikichi Y,et al. Mobilization of CD34-positive bone marrow-derived cells after coronary stent implantation: impact on restenosis. Circulation. 2007;115(5):553-561.[12] Liu KQ, Qi X, Du JP,et al. Treatment of acute myocardial infarction with autologous bone marrow stem cells mobilization combined with recombinant growth factor in rat. Zhongguo Wei Zhong Bing Ji Jiu Yi Xue. 2006;18(8):494-497.[13] Nikol S, Kuhlmann M. Mobilization of stem cells in cardiovascular disease. Improvement of vascularization, cardiac function and arrhythmias.Med Klin (Munich). 2006; 22:190-192.[14] Kanellakis P, Slater NJ, Du XJ, et al. Granulocyte colony-stimulating factor and stem cell factor improve endogenous repair after myocardial infarction .Cardiovasc Res. 2006;70(1):117-125. [15] Jakob P, Landmesser U. Current status of cell-based therapy or heart failure. Curr Heart Fail Rep. 2013;10(2):165-176.[16] Koninckx R, Daniëls A, Windmolders S, et al. The cardiac atrial appendage stem cell: a new and promising candidate for myocardial repair. Cardiovasc Res. 2013;97(3):413-423.[17] Honold J, Fischer-Rasokat U, Lehmann R,et al. G-CSF stimulation and coronary reinfusion of mobilized circulating mononuclear proangiogenic cells in patients with chronic ischemic heart disease:five-year results of the TOPCARE-G-CSF trial. Cell Transplant. 2012;21(11): 2325-2337.[18] Mozid AM, Jones D, Arnous S, et al. The effects of age, disease state, and granulocyte colony-stimulating factor on progenitor cell count and function in patients undergoing cell therapy for cardiac disease. Stem Cells Dev. 2013;22(2): 216-223.[19] Konenkov VI, Pokushalov EA, Poveshchenko OV, et al. Phenotype of peripheral blood cells mobilized by granulocyte colony-stimulating factor in patients with chronicheart failure. Bull Exp Biol Med. 2012;153(1):124-128.[20] Ripa RS. Granulocyte-colony stimulating factor therapy to induce neovascularization in ischemic heart disease. Dan Med J. 2012;59(3):B4411. [21] Zimmet H, Porapakkham P, Porapakkham P, et al. Short- and long-term outcomes of intracoronary and endogenously mobilized bone marrow stem cells in the treatment of ST-segment elevation myocardial infarction: a meta-analysis of randomized control trials. Eur J Heart Fail. 2012;14(1): 91-105.[22] Zhao Q, Sun C, Xu X, et al. CD34+ cell mobilization and upregulation of myocardial cytokines in a rabbit model of myocardial ischemia. Int J Cardiol. 2011;152(1):18-23.[23] Ichim TE, Solano F, Lara F, et al. Combination stem cell therapy for heart failure. Int Arch Med. 2010;3(1):5. [24] Hou M, Hu SS, Wei YJ, et al. Effects of mobilization of bone marrow stem cells on regenerative potential of cardiac tissues after myocardial infarction. Zhonghua Yi Xue Za Zhi. 2009; 89(17):1192-1196.[25] 姚巍,王凤芝.骨髓间充质细胞静脉移植联合自体骨髓干细胞动员对心肌病心力衰竭大鼠的心功能影响[J].中国组织工程研究与临床康复,2008,12(29):5645-5648.[26] 姚巍,王凤芝.重组人生长激素联合骨髓间充质干细胞静脉移植对心力衰竭大鼠心功能的影响[J].中国药物与临床,2007,7(11): 825-827.[27] Helmuth L. Stem cell shear call of injured tissue.Science. 2000; 290: 1479-1481.[28] Dotsenko O, Xiao Q, Xu Q, et al. Bone marrow resident and circulating progenitor cells in patients undergoing cardiac surgery. Ann Thorac Surg. 2010;90(6):1944-1951.[29] Habeeb NM, Youssef OI, El Hadidi ES. Therapeutic role of mobilized bone marrow cells in children with nonischemic dilated cardiomyopathy. ISRN Pediatr. 2012;2012:927-968.[30] Druka?a J, Paczkowska E, Kucia M, et al.. Stem cells, including a population of very small embryonic-like stem cells, are mobilized into peripheral blood in patients after skin burn injury. Stem Cell Rev. 2012;8(1):184-194.[31] Wright DE, Wagers AJ, Gulati AP, et al.Physiological migration of hematopoietic stem and progenitor cells. Science. 2001;294: 1933-1936.[32] OrlicD,Kajstura J,Chimenti S,et al. Bone marrow cells regenerate infracted myocardium. Nature. 2001;410(6829): 701-705.[33] Stroncek DF, Clay ME, Herr G, et al. The kinetics of G-CSF mobilization of CD34+ cells in healthy people. Transfus Med. 1997;7:19-24.[34] Harada M, Nagafuji K, Fujisaki T, et al. G-CSF-induced mobilization of peripheral blood stem cells from healthy adults for allogeneic transplantation. J Hematother. 1996;5(1):63-71.[35] Kocher AA, Schuster MD, Szabolcs MJ, et al. Neovascularization of ischemic myocardium by human bone-marrow-derived angioblasts preventscardiomyocyte apoptosis, reduces remodeling and improves cardiac function. Nat Med. 2001;7: 430-436.[36] Ciulla MM, Lazzari L, Pacchiana R, et al. Homing of peripherally injected bone marrow cells in the rat after experimental myocardial injury. Haematologica. 2003;88: 614-621.[37] Stroncek DF, Clay ME, Herr G, et al. The kinetics of G-CSF mobilization of CD34+ cells in healthy people. Transfus Med. 1997;7:19-24.[38] Kajstura J, Zhang X, Reiss K, et al. Myocyte cellular hyperplasia and Myocyte cellular hypertrophy contribute to chronic ventricular remodeling in coronary artery narrowing -induced cardiommyopathy in rats. Circ Res. 1994; 74(3): 383-400.[39] 欧瑞明,陈运贤,钟雪云,等.粒细胞集落刺激因子对大鼠实验性心肌梗死治疗作用[J].广东医学,2002,23(11):1131-1133.[40] 刘宏伟,盖鲁粤,张端珍,等.粒细胞集落刺激因子骨髓干细胞动员治疗免心肌梗死的实验研究[J].军医进修学院学报,2005, 26(2): 132-134.[41] Orlic D, Kajstura J, Chimenti S, et al. Mobilized bone marrow cells repair the Infracted heart, improving function and survival. Proc Natl Acad Sci. 2001;98:10344-10349. |
| [1] | Wang Jian-ji, Yang Long, Li Jing, Sun Qi, Zuo Wei-min, Ren Qi-feng, Sun Yu, Wu Zhan-yu, Zou Qiang, Ma Min-xian, Ye Chuan. Development and application of special-purpose grafter by femoral head decompression combined with bone marrow mesenchymal stem cells transplantation based on three-dimensional printing technology [J]. Chinese Journal of Tissue Engineering Research, 2016, 20(44): 6636-6642. |
| [2] | Zhou Chang-yan, Zhou Qing-huan, Bian Jing, Chen Ke, Chen Wen. Bone marrow mesenchymal stem cells combined with calcium phosphate cement to repair articular cartilage defects in rabbits [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(8): 1195-1199. |
| [3] | Du Qing-hua, Cao Jun-kai, Dong Xi-xi, E Ling-ling, Wei Li-jun. Osteogenic differentiation of pluripotent stem cells induced by akermanite extracts [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2236-2242. |
| [4] | Wu Yan, Huang Lan . Bone morphogenetic protein 9-induced osteogenic differentiation of dental follicle cells in vitro [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2255-2260. |
| [5] | Rao Li-jia, Li Qi-meng, Li Jin-ling, Xu Qiong. Expression pattern of ten-eleven translocation family during differentiation of human dental pulp cells [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2261-2266. |
| [6] | Gao Zhuo-yue, Liu Yong-qi, He Jian-xin, Wu Zhi-wei, Luo Ya-li, Su Yun, Zhang Li-ying, Zhang Qi, Wu You-ming, Zhou Ni-na. Regulatory effects of warming yang and invigorating qi treatment on the inflammatory balance and genetic stability of bone marrow mesenchymal stem cells under tumor microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2267-2272. |
| [7] | Wang Fang, Chen Shao-wei. In vitro culture of embryos and establishment of embryonic stem cell lines [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2273-2277. |
| [8] |
Du Wei-bin, Quan Ren-fu, Zheng Xuan, Wang Tuo.
Hair follicle stem cells promote the healing of skin wound
|
| [9] | Du Hua, Shi Ying-xu . Bone marrow microenvironment controls the “fate” of hematopoietic stem cells [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2283-2290. |
| [10] | Xu Xiang, Yin He-ping. Platelet-rich plasma accelerates the proliferation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2144-2148. |
| [11] | Ma Fa-ku, Wang Huan, Liu Bin, Yang Yan-li, Su Qin-jun, Qian Zhen, Dong Liang. Expression of CD44+/C-myc+ cancer stem cells and its relationship with the prognosis of patients in colorectal tumors [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2161-2166. |
| [12] |
Chen Ping, Wang Jian.
Enrichment of lung cancer stem cells and expression of related markers
[J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2167-2171.
|
| [13] | Fan Zhi-gang, Fan Hong-liang. Oxidative stress response in diabetic nephropathy rats following injection of embryonic stem cells via the tail vein [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2199-2204. |
| [14] | Wang Yong, Zhao Wei, Feng Jian-zhou, Chen Xiao-chun. Nerve growth factor-modified adipose derived stem cells for repair of spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2224-2229. |
| [15] | Sha Wen-qiong, She Rui-lian, Wang Zi-neng, Ke Ru. Ultrastructure and phagocytotic function of human placental mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(14): 2230-2235. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||